Japan Begins Ebola Vaccine Candidate Tests
A research team at the University of Tokyo announced ‘they will begin a clinical study on a vaccine candidate for the Ebola virus, a first in Japan.’
This Ebola vaccine candidate was developed using an artificially produced detoxified virus, commented Yoshihiro Kawaoka, Ph.D., a professor at the university's Institute of Medical Science, in news reports on December 5, 2019.
Dr. Kawaoka developed the new vaccine using an inactivated Ebola virus that cannot replicate in normal cells. Although possession of the Ebola virus is strictly regulated in Japan, artificially produced viruses are not.
This Ebola vaccine was proven both safe and effective in tests involving monkeys.
This experimental Ebola vaccine, developed in the lab of UW-Madison scientist Yoshihiro Kawaoka, was produced at Waisman Biomanufacturing, a specialized center at the university’s Waisman Center.
Unlike other experimental vaccines, the vaccine created by Kawaoka and Halfmann does not rely on a secondary, live-though-weakened virus to deliver a portion of the Ebola virus to the human immune system.
The UW-Madison version, considered a “whole-virus” vaccine, uses 7 of the 8 genes that make up the Ebola virus. The UW-Madison vaccine is missing a key protein and can grow only in special cells engineered to contain the protein, the university said in a press release on September 30, 2019.
The form of Ebola virus contained in the vaccine is a technology Halfmann created more than a decade ago, called DeltaVP30.
This technology renders the virus incapable of reproducing itself because it eliminates a gene that makes a protein critical for this task. The virus can only grow in a special cellular system containing the missing protein, which is not found in human or animal cells. Prior to use in humans, it was proven safe and effective in mice, guinea pigs, and in non-human primates.
Other Ebola vaccines use vector viruses to deliver a single Ebola protein to the immune system.
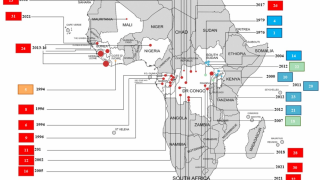
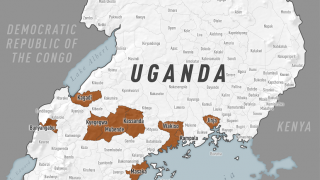
The researchers said they aim to develop the Ebola vaccine to prevent further outbreaks of the deadly hemorrhagic fever in Africa.
Furthermore, this vaccine is believed by these researchers to have fewer side effects compared to other vaccines produced abroad.
The clinical study in Japan will be conducted by the team led by professor Hiroshi Yotsuyanagi at the hospital affiliated with the Institute.
Beginning in December 2019, about 30 healthy adult men are set to receive intramuscular injections of the vaccine and will be checked for fevers as well as the development of antibodies.
They will then be monitored for about 6 months to determine the vaccine's safety.
There are 2 Ebola vaccines currently being tested in Africa.
- Merck’s Ervebo (V920) is a recombinant, replication-competent Ebola vaccine
- Jannsen’s Ad26.ZEBOV/MVA-BN a heterologous prime-boost Ebola vaccine regimen.
Ebola vaccine news is published by Precision Vaccinations
Our Trust Standards: Medical Advisory Committee