Ebola Vaccine Candidate Reported 96% Seroconverted after 3 Doses

The Ebola vaccine candidate INO-4201 was reported safe, tolerable, and generated strong T cell and antibody responses in a small clinical trial published on March 21, 2019.
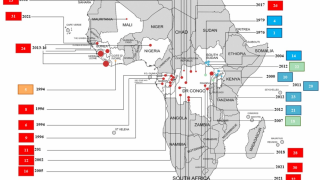
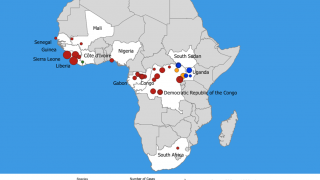
This is important news since there is not a commercially available Ebola vaccine available today.
Of 70 evaluated Phase 1 study subjects, 67 (96%) seroconverted and mounted a strong antibody response to the Ebola glycoprotein antigen following the 3 dose immunization regimen; 52 subjects (76%) seroconverted after only 2 doses.
Significantly, in the study arm using intradermal (skin) administration, 13 of 13 evaluable subjects (100%) generated antigen-specific antibody responses after only 2 doses and all remained seropositive after three immunizations.
Unlike viral vector vaccines which must be kept frozen, INO-4201 is reported stable at room temperature for more than 1 year. Non-live vaccine approaches that are simple to deliver and stable at room temperature are desirable in controlling Ebola virus outbreaks in countries without appropriate infrastructure.
This Phase 1 study demonstrated that intradermal (skin) administration with INO-4201 generated T cell responses equivalent to or better than the group that received intramuscular delivery.
Dr. J. Joseph Kim, Inovio's President and CEO said in a press release, "Inovio's Ebola vaccine candidate INO-4201 can be used to protect against Ebola infections and can be boosted multiple times without any anti-vector response, it could be employed to boost viral vector vaccines that cannot be effectively re-administered."
To date, INO-4201 has not demonstrated systemic serious adverse effects, such as fever, joint pain, and low white blood cell counts, reported in association with some viral vector-based Ebola vaccines currently in development.
This study was fully funded by the U.S. Defense Advanced Research Projects Agency (DARPA) and can be found in the most recent edition of The Journal of Infectious Diseases in the article entitled, "Intradermal SynCon® Ebola GP DNA Vaccine is Temperature Stable and Safely Demonstrates Cellular and Humoral Immunogenicity Advantages in Healthy Volunteers," authored by Inovio and its collaborators.
At present, there are no FDA-approved pre- or post-exposure interventions available in the event of an Ebola outbreak, laboratory accident, or deliberate misuse.
However, Merck’s Ebola vaccine candidate v920 has been administered to 87,900 people in several countries in Africa during 2018-2019.
And on February 20, 2019, the v920 vaccine was authorized for pregnant and lactating women in Africa.
Inovio Pharmaceuticals, Inc. is a late-stage biotechnology company focused on the discovery, development, and commercialization of DNA immunotherapies that transform the treatment of cancer and infectious diseases.
Our Trust Standards: Medical Advisory Committee
- Intradermal SynCon® Ebola GP DNA Vaccine is Temperature Stable and Safely Demonstrates Cellular and Humoral Immunogenicity
- Open-Label Study of INO-4212 With or Without INO-9012, Administered IM or ID Followed by Electroporation in Healthy Volunteers
- Inovio's Ebola Vaccine Delivered Intradermally Demonstrates 100% Immunogenicity in a Clinical Study
- Inovio Zika vaccine candidate INO-A002 launches phase 1 study