Should Kentucky and Tennessee Require Hepatitis A Vaccinations for Food Staff?

The expanding hepatitis A outbreak in 'Heartland' states may be sourced from food service staff.
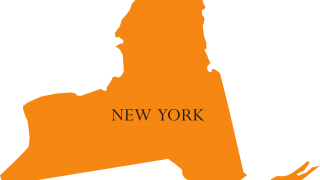
On June 2, 2018, the Kentucky Department of Health (KDH) reported 761 hepatitis A cases, which include 467 hospitalizations and 6 related fatalities. But, only 502 of these 761 people are the typical drug user or homeless person.
The Centers for Disease Control and Prevention (CDC) national risk factor report for June 11, 2018, identified 68 percent of the infected used drugs, were homelessness, or both.
This means, 32 percent of hepatitis A cases are diagnosed in other people, such as food service staff.
The CDC says food service workers with hepatitis A can transmit the virus to customers by contaminating surfaces, utensils, or food.
Since hepatitis A symptoms appear 15 to 50 days after the initial exposure, the food handler is generally not ill during the peak time of infectivity which occurs two weeks before illness begins, reports Food Safety News.
As an example in Kentucky, on June 20, 2018, WSAZ reported, officials with the Ashland-Boyd County Health Department confirmed a 10th food service worker has been diagnosed with hepatitis A.
Boyd County officials say a restaurant employee was diagnosed and the restaurant voluntarily closed to complete an extensive disinfection of the facility.
But, this WSAZ news report did not mention if the infected restaurant employee was furloughed, to limit future infections.
Which means, the Kentucky state-wide hepatitis A outbreak may continue in an endless loop.
Tennessee may also be in an endless outbreak loop.
In Nashville, TN on June 20, 2018, the Department of Health issued a press release, “We are moving forward in our campaign to reach as many as possible among the at-risk population,” said Rachel Franklin, Interim Director of Communicable Disease and Emergency Preparedness.
“The outbreak will likely last for a number of months, so we will continue to respond with a long-term vaccination effort to prevent the spread of the virus.”
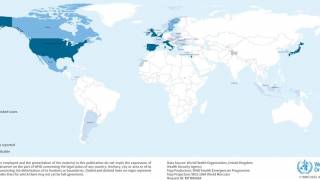
In response to these cases, on June 11, 2018, the CDC issued an official Health Advisory regarding the ongoing hepatitis A outbreaks. During 2018, more than 2,500 hepatitis A infections have been reported associated with person-to-person transmission.
These hepatitis A cases have been reported in various states, primarily clustered in the Midwest.
Other states have also reported hepatitis A cases in food service staff:
According to the CDC, the best way to prevent hepatitis A virus (HAV) infection is through vaccination. The number and timing of the doses depend on the type of vaccine administered.
The inactivated vaccines containing HAV antigen currently licensed in the United States are:
- the single-antigen vaccines HAVRIX® (manufactured by GlaxoSmithKline, Rixensart, Belgium) and,
- VAQTA® (manufactured by Merck & Co., Inc., Whitehouse Station, New Jersey) and,
- the combination vaccine TWINRIX® (containing both HAV and hepatitis B virus antigens; manufactured by GlaxoSmithKline).
- GamaSTAN S/D (Grifols Therapeutics, Inc., Research Triangle Park, North Carolina) immune globulin (IG) for intramuscular administration is the only IG product approved for HAV prophylaxis.
The efficacy of IG or vaccine, when administered greater than 2 weeks after exposure, has not been established, says the CDC.
Most pharmacies in the USA offer these hepatitis vaccines.
To schedule a vaccination appointment at a local pharmacy, please click here.
The CDC Vaccine Price List provides current HAV vaccine contract prices and general information, and vaccine discounts can be found here.
Vaccines, like any medicine, can have side effects, says the CDC. You are encouraged to report negative side effects of vaccines to the FDA or CDC.
Our Trust Standards: Medical Advisory Committee
- Kentucky Department for Public Health KY17-089 - Acute Hepatitis A Outbreak Weekly Report
- Foodservice workers should be vaccinated against Hepatitis A
- Outbreak of Hepatitis A Virus (HAV) Infections among Persons Who Use Drugs and Persons Experiencing Homelessness
- Should Kentucky Derby Food Staff Be Vaccinated for Hepatitis A?
- Hepatitis A Health Advisory Issued by CDC
- INFECTIOUS DISEASE BRANCH Hepatitis A
- Hepatitis case at Callihan’s