Innovative TB Vaccine Found 49.7% Effective

Tuberculosis (TB) vaccine candidate GSK’s M72/AS01 was reported to significantly reduced the incidence of pulmonary TB disease in HIV-negative adults with latent TB infection.
This phase 2b study’s results demonstrated an overall vaccine efficacy of 49.7 percent during the 3-years after vaccination.
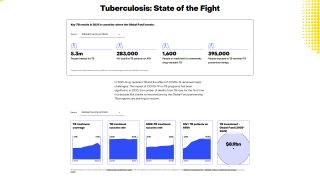
This vaccine efficacy level is lower than for most approved vaccine, however, these health experts contended that, with 10.4 million new TB cases and 1.7 million fatalities each year, the number of lives saved from this TB vaccine would be great.
Moreover, multi-drug resistant strains of TB are emerging and spreading, and the only available TB vaccine, BCG, does not provide proven and consistent protection in adults in TB-endemic countries.
The clinical trial was conducted in TB-endemic Kenya, South Africa, and Zambia and involved 3,573 HIV-negative adults between the ages of 18 and 50 years. Participants who received 2-doses of either M72/AS01E or placebo 30 days apart were followed for 3-years to detect evidence of pulmonary tuberculosis disease.
In the final analysis, 13 participants in the vaccine group developed active pulmonary TB compared to 26 participants in the placebo group.
Among the study participants who received the vaccine, the increased M72-specific immune response was sustained through 3-years.
The final results are consistent with the primary analysis done after 2-years of follow-up and published in the New England Journal of Medicine in September 2018.
Dr. Thomas Breuer, Chief Medical Officer of GSK Vaccines, said in a press release, “These results demonstrate that for the first time in almost a century, the global community potentially has a new tool to help provide protection against TB.”
GSK’s M72/AS01E candidate vaccine contains the M72 recombinant fusion protein, derived from two Mycobacterium tuberculosis antigens (Mtb32A and Mtb39A), combined with the Adjuvant System AS01, which is also a component of GSK’s RTS,S malaria vaccine and vaccine against shingles, Shingrix.
An effective vaccine against tuberculosis administered in adolescents and adults would have a marked impact on tuberculosis control, including drug-resistant tuberculosis, through interruption of transmission, and it would help achieve the WHO target of ending the tuberculosis epidemic by 2035, said GSK.
It is estimated that 25 percent of the global population has latent TB infection, of whom, approximately 10 percent will develop active pulmonary TB disease.
Recent TB vaccine news
- Rapid Triage Test for Tuberculosis Identified
- BCG Vaccination Reduced Lung Cancer Risks By 2.5-Fold
- An Old Vaccine Keeps Getting Better
- Diagnosing Latent Tuberculosis Infection
The study was sponsored by GSK and conducted in partnership with IAVI. Funders of IAVI for this study were the Bill & Melinda Gates Foundation, the United Kingdom’s Department for International Development, the Directorate-General for International Cooperation in the Netherlands, and the Australian Agency for International Development.
IAVI is a nonprofit scientific research organization dedicated to addressing urgent, unmet global health challenges including HIV and tuberculosis. Our mission is to translate scientific discoveries into affordable, globally accessible public health solutions. Read more at IAVI.
GSK is a science-led global healthcare company with a special purpose: to help people do more, feel better, live longer. For further information please visit GSK.
Tuberculosis vaccine news published by Precision Vaccinations
Our Trust Standards: Medical Advisory Committee
- Final Analysis of a Trial of M72/AS01E Vaccine to Prevent Tuberculosis List of authors.
- Study to Evaluate the Efficacy of GlaxoSmithKline (GSK) Biologicals' Candidate Tuberculosis (TB) Vaccine in Adults
- GSK candidate vaccine demonstrates sustained level of protection against active pulmonary tuberculosis