Flu Shots Flopped Last Season
During the two-day U.S. CDC Advisory Committee on Immunization Practices (ACIP) meeting led by Dr. Grace Lee, several presentations revealed significant vaccine progress and measurable health impact.
However, when Jessie Chung MPH, Samantha Olson MPH, and Nathaniel Lewis Ph.D. presented the preliminary influenza vaccine efficacy (VE) results from the last flu season, various ACIP members expressed shock and dismay.
On slide (#18), the summary data revealed for the 2021–2022 influenza season, vaccinations provided low to non-significant protection against predominant influenza A/H3N2 illness of varying severity.
This presentation consolidated data from three networks to evaluate vaccine effectiveness against laboratory-confirmed influenza-associated outpatient visits, emergency department visits, and hospitalization.
The data suggested higher VE in outpatient settings compared to pediatric hospitalization/ED visits but not adult inpatient influenza.
The CDC staff presented the preliminary VE data as follows:
- 36% (95%CI: 20, 49) against outpatient illness aged ≥6 months (Flu VE),
- 31% (95%CI: -14, 58) against pediatric hospitalizations (NVSN),
- 19% (95%CI: -12, 41) against pediatric ED visits (NVSN),
- 11% (95%CI: -19, 33) against adult hospitalizations (IVY).
In addition, this new data indicates influenza protection was similar to pre-pandemic A/H3N2-dominant seasons.
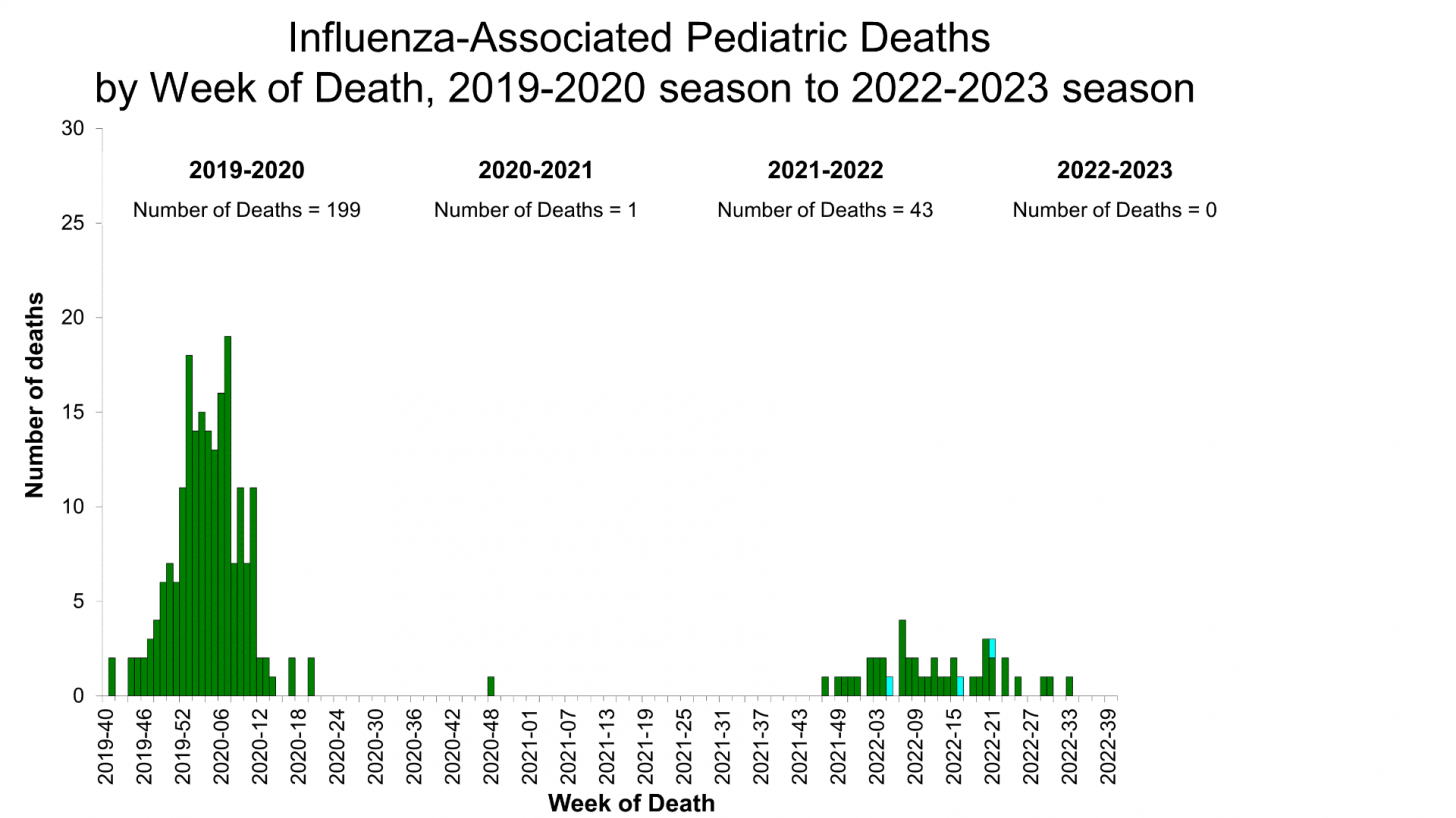
The ACIP members focused their comments on the pediatric VE since the CDC recently reported the 43rd influenza-associated pediatric deaths that occurred during the 2021-2022 season.
This report highlighted that only one fatality was reported during the 2020-2021 flu season and 199 during the 2019-2020 season.
As the ACIP members debated flu shot selection mismatch possibilities with the previous circulating influenza viruses, CDC staff reminded the committee that influenza A(H3N2) viruses remain predominant in the U.S. and the Southern Hemisphere.
The CDC confirmed on June 22, 2022, that 2022–23 Northern Hemisphere influenza vaccines would include updated reference viruses for A(H3N2) (clade 3C.2a1b, subclade 2a.2) and B/Victoria (B/Austria/1359417/2021).
As the U.S. is now reporting influenza outbreaks, Lynnette Brammer, MPH, presented information regarding Australia's just-ending flu season.
Australia reported an early and shorter 2022 influenza season compared to historical trends.
In 2022, 99.7% of specimens have been influenza A, with H3 making up 91.3% of subtyped specimens.
Unfortunately, the Department of Health and Aged Care recently reported that flu shot VE is on the "lower end of the moderate range" from preliminary estimates in 2022.
The ACIP members closed off the flu shot presentation with a consensus that the potential for co-circulation of influenza, COVID-19, pneumonia, and other respiratory viruses could add substantial pressure this winter.
The CDC believes influenza vaccination can reduce illnesses, even at reduced VE.
Throughout the U.S., various flu shots are available at most clinics and local pharmacies, generally at no or minimal cost to consumers.
Additional flu shot news for the 2022-2023 season is posted at PrecisionVaccinations.com/Flu.
PrecisionVaccinations publishes fact-checked, research-based vaccine information manually curated for mobile readership.
Our Trust Standards: Medical Advisory Committee
























