Chikungunya Vaccine CHIKV-VLP Gains PRIME Designation in Europe

A Maryland based life science company announced that its Chikungunya Virus virus-like particle (VLP) vaccine candidate, CHIKV VLP, was granted PRIority MEdicines, or PRIME, designation by the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA).
Emergent BioSolutions’ CHIKV VLP vaccine candidate is being developed for the prevention of disease caused by a chikungunya virus infection, for which no vaccine or treatment is currently available.
The PRIME scheme is designed to enhance support for the development of medicines that target an unmet medical need and that may offer a major therapeutic advantage over existing treatments, or benefit patients without preventative or treatment options.
“Emergent is pleased to receive this important EMA designation and encouraged by the strength of our early data package, including results from the interim analysis of our Phase 2 safety and immunogenicity study for our CHIKV VLP vaccine candidate announced last April,” said Abbey Jenkins, senior vice president, in a press release.
That phase 2 study found the CHIKV-VLP vaccine candidate produced 74 to 98 percent seroconversion across a series of dosing regimens.
Previous studies have shown that VLP vaccines are highly immunogenic, have a proven safety record, and typically elicit high titer neutralizing antibodies needed to protect against the chikungunya virus.
“We look forward to working with the EMA to execute on our development plan, which contemplates initiation of a pivotal (clinical) trial in 2020,” said Jenkins.
Under the PRIME scheme, eligible companies can benefit from enhanced interaction and early dialogue with the EMA to help optimize development plans and expedite evaluation such that medicines under development can reach patients faster.
The CHIKV VLP vaccine candidate is licensed from the National Institute of Allergy and Infectious Diseases at the National Institutes of Health. It received Fast Track designation from the U.S. Food and Drug Administration in May 2018.
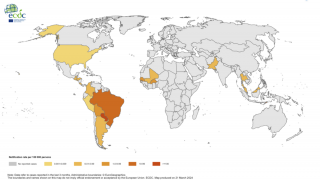
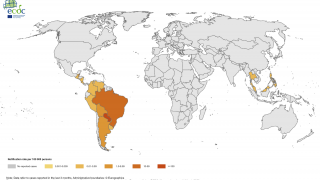
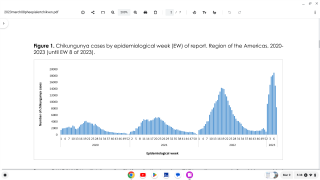
Chikungunya virus outbreaks have occurred in countries in Africa, Asia, Europe, and the Indian and Pacific Oceans.
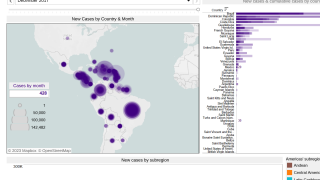
Chikungunya virus disease cases have recently been reported in Ethiopia, Brazil, Thailand, and Honduras. At the end of July 2019, both Suriname and Argentina reported their first case of chikungunya, says the European CDC.
The Chikungunya virus was found for the first time in the Americas on various islands in the Caribbean in 2013, says the US Centers for Disease Control and Prevention (CDC).
As of October 3, 2019, a total of 69 chikungunya virus disease cases with illness onset in 2019 have been reported from 23 U.S. states. During 2018, a total of 90 chikungunya virus disease cases were confirmed in the USA.
All of these reported cases occurred in travelers returning from affected areas.
This means there is an ‘ongoing risk that the virus will be imported to new areas by infected international travelers.’
No locally-transmitted cases have been reported from U.S. states, says the CDC. But, there have been 2 locally acquired chikungunya cases reported from Puerto Rico in 2019.
Chikungunya virus is spread to people by the bite of an infected mosquito. The most common symptoms of infection are fever and joint pain. Other symptoms may include headache, muscle pain, joint swelling, or rash, says the CDC.
Emergent BioSolutions Inc. is a global life sciences company seeking to protect and enhance life by focusing on providing specialty products for civilian and military populations that address accidental, deliberate, and naturally occurring public health threats.
Chikungunya vaccine news published by Precision Vaccinations
Our Trust Standards: Medical Advisory Committee