New Dengue Virus Test Needed

Recent safety concerns are threatening the global dengue virus vaccine effort.
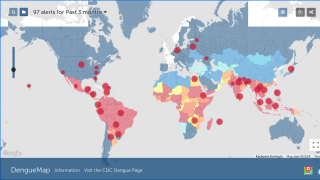
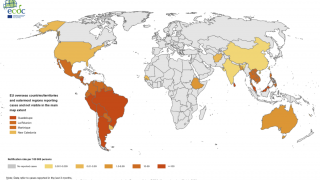
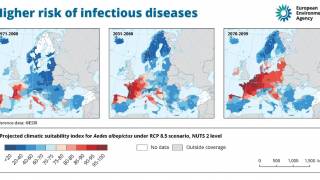
The diagnosis of arboviruses has been a challenge for decades as dengue viruses have expanded their global reach.
Recently, new research has linked the new dengue vaccine, Dengvaxia, to false-positive test results.
Which has led to a start / stop launch of the Dengvaxia vaccine.
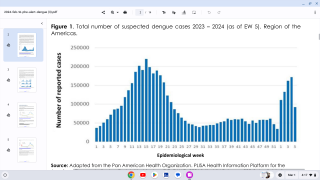
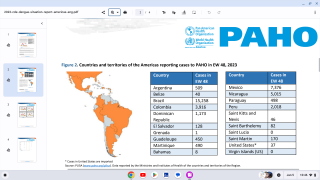
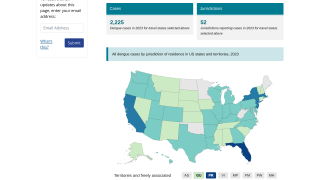
This confusion is a setback for scientists who have spent decades developing a vaccine against dengue, which is a mosquito-borne virus that infects an estimated 390 million people worldwide annually.
Two phase 3 clinical trials have produced data that indicates that the Dengvaxia vaccine impacts the efficacy of immunoglobulin M- and immunoglobulin G-based assays, which test for dengue virus infection.
These researchers believe their findings identify the need for new dengue-specific diagnostics. They found evidence that Dengvaxia-induced responses may impact the results from assays that use IgM and IgG data to detect infection.
According to the researchers, IgM- and IgG-based assays led to a higher number of false-positive results among vaccinated participants.
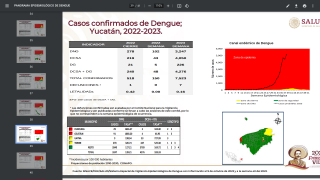
The assays yielded a negative predictive value of 99.5 percent and a positive predictive value of just 22.9 percent.
“The data from all four serotypes, allowed us to explore the impact of previous dengue exposure and other potential covariates on the serological diagnosis of dengue,” the researchers noted.
In summary, this research indicates the urgent need for new assays that are not affected by vaccine-induced immunity.
Dengvaxia is a live, recombinant, tetravalent vaccine that is delivered in three doses, 6 months apart. According to World Health Organization (WHO), it is the first licensed dengue vaccine and has been approved for use in endemic areas.
The Dengvaxia vaccine was introduced in vaccination programs during 2017 in the Philippines and Brazil.
These immunization programs were based upon data from clinical trials which showed the vaccine reduced severe dengue by 93% and hospitalizations by 81% within the first 25 months of vaccination, according to WHO.
However, participants aged 2 to 5 years showed an increased risk for hospitalization. These participants were 5 times more likely to be hospitalized if they had a prior dengue virus infection.
These researchers disclosed various relationships with the pharma industry.
Our Trust Standards: Medical Advisory Committee
- DENGVAXIA®, WORLD’S FIRST DENGUE VACCINE, APPROVED IN MEXICO
- Impact of Dengue Vaccination on Serological Diagnosis: Insights From Phase III Dengue Vaccine Efficacy Trials
- Arbovirus Diagnostics: From Bad to Worse due to Expanding Dengue Virus Vaccination and Zika Virus Epidemics
- Immunogenicity and safety of one versus two doses of tetravalent dengue vaccine in healthy children aged 2–17 years in Asia and