Chikungunya Virus Effect Found to Be Long Lasting

The Chikungunya outbreak continues to expand around the world, and researchers are learning more how this virus impacts people.
In a retrospective cohort study published in Travel Medicine and Infectious Disease, researchers found that chronic inflammatory rheumatism is common in patients in Colombia and Latin America, even a full year after infection.
Scientists from Colombia reported that 58 percent of lab-confirmed chikungunya cases had at least one rheumatologic symptom one year after the initial infection.
These results are consistent with results from other studies in France. Chikungunya has been identified in 60 countries by the World Health Organization.
Italy's chikungunya outbreak earned a Centers for Disease Control and Prevention (CDC) Travel Health Watch - Level 1 in October 2017.
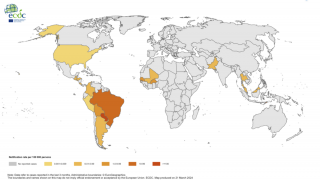
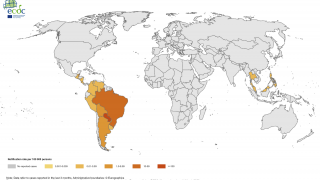
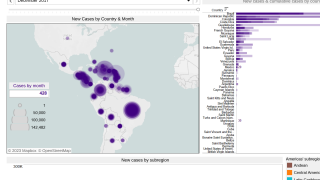
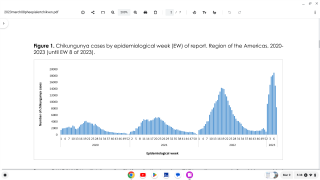
In the Pan American Health Organization (PAHO) region, 1,106 new chikungunya infections were reported during November 2017.
This increase brings the PAHO yearly total to 184,636 confirmed, suspected, and imported cases.
As of late November 2017, a total of 92 chikungunya virus disease cases with illness onset in 2017 have been reported from 23 U.S. states to the CDC.
All reported chikungunya cases occurred in travelers returning from affected areas.
Which means, no locally-transmitted cases have been reported from U.S.A.
But, these updated totals may not tell the entire PAHO situation.
Brazil, which has reported more than 90% of this year's total, has not reported its updated number of chikungunya cases to PAHO for months.
The chikungunya virus is transmitted from human to human by the bites of infected female Aedes albopictus mosquitoes.
Chikungunya virus symptoms include fever, headache, nausea, vomiting, rash, and pain in the eyes, joints, and muscles. Chikungunya virus also shares some clinical signs with dengue, and can be easily misdiagnosed, says the CDC.
There is not an FDA approved vaccine to protect people from the chikungunya virus.
A prophylactic vaccine candidate against the chikungunya fever was scheduled for phase II clinical study in Puerto Rico before Hurricane Maria struck.
According to the vaccine manufacturer, this vaccine candidate already showed high seroconversion rates in the preceding phase I clinical trial. Up to 100 percent of all vaccinated subjects produced neutralizing antibodies against the chikungunya virus.
Our Trust Standards: Medical Advisory Committee
- Post-chikungunya chronic inflammatory rheumatism: Follow-up of cases after 1 year of infection in Tolima, Colombia
- Innovate UK has granted Themis £3M (€3.3M) to push its Chikungunya Vaccine towards Phase III.
- Chikungunya Outbreak in Italy Earns CDC Travel Alert
- Chikungunya Virus
- Pan American Health Organization