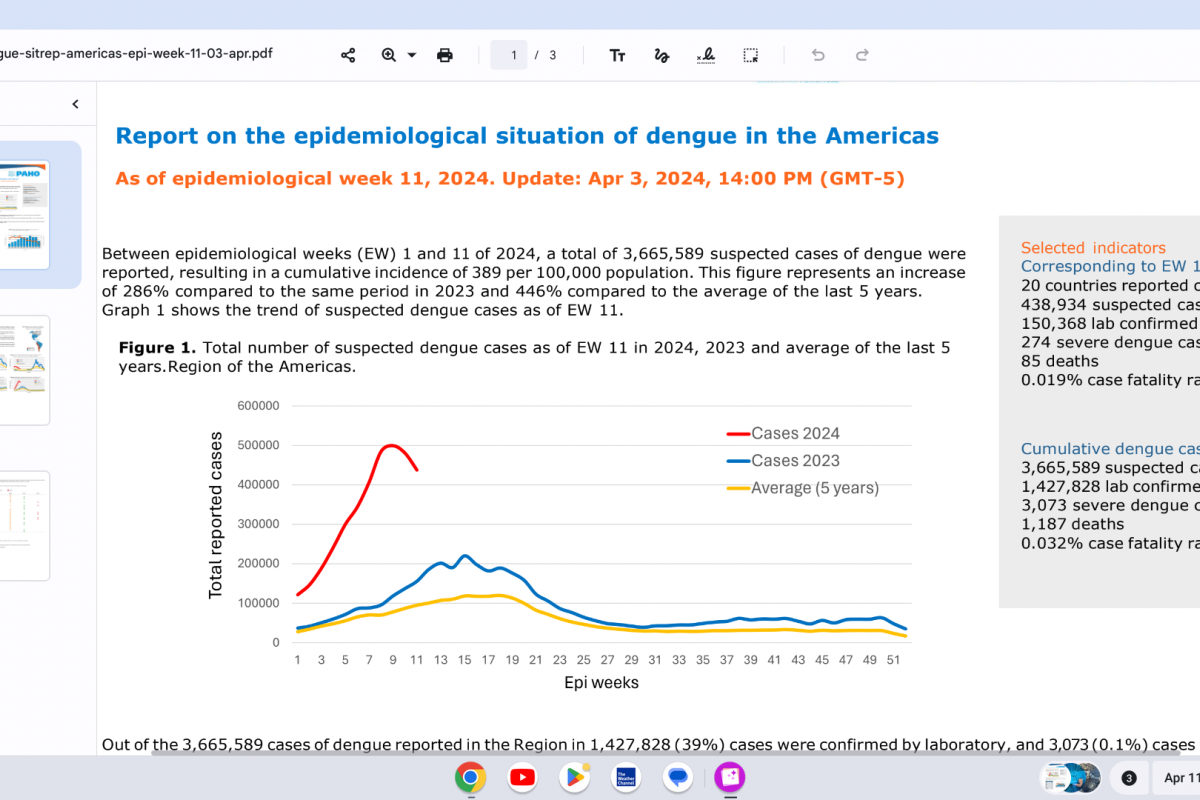
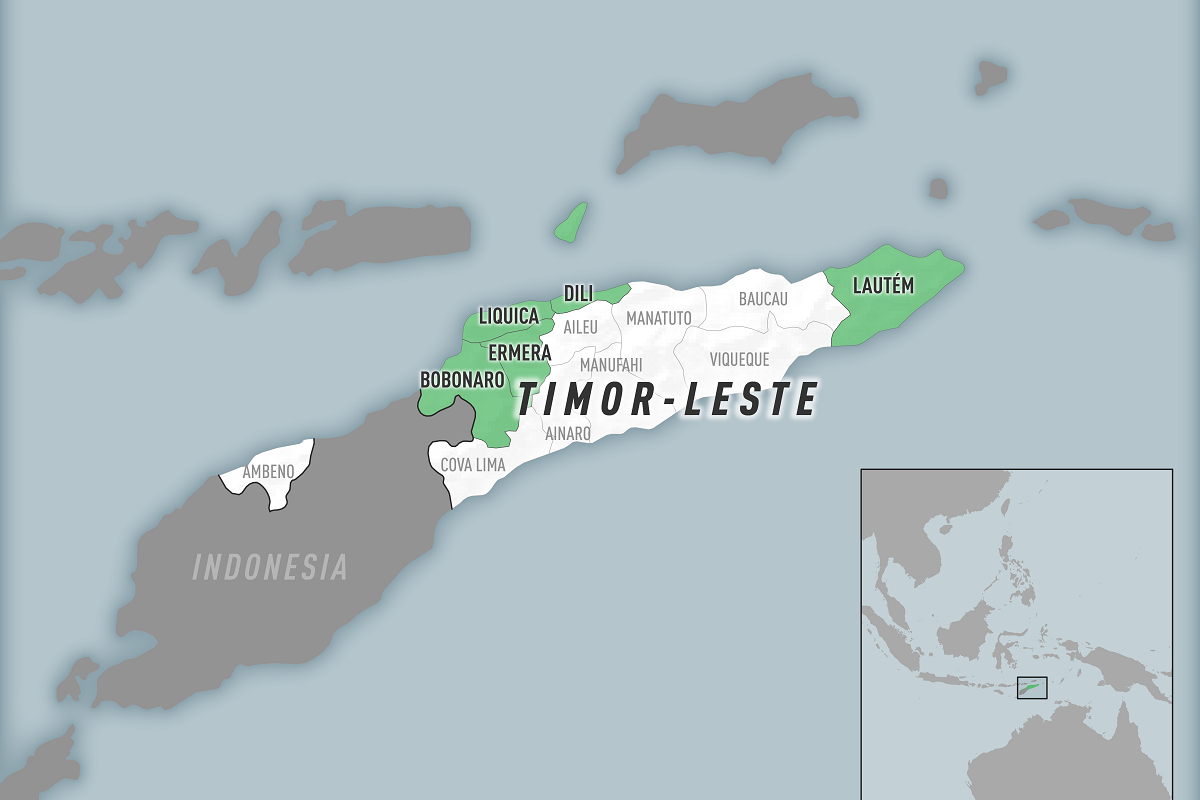
A multi-month chikungunya virus outbreak continues impacting the residents of Timor-Leste (East Timor). As of late March 2024, 195 chikungunya cases have been reported in Timor-Leste.
The U.S. CDC reissued a Level 2 - Practice Enhanced Precautions Travel Health Advisory on April 5, 2024, to alert international travelers. Data sources indicate tourist arrivals in East Timor increased to 3,718 in the first quarter of 2022.
Additionally, neighboring countries, such as Thailand (139), Pakistan (26), and Malaysia (6), reported chikungunya cases in 2024.
The Chikungunya virus causes this mosquito-transmitted disease. Most people infected develop some symptoms, but related fatalities are rare.
However, people at risk for more severe disease include newborns infected around the time of birth, older adults, and people with medical conditions such as high blood pressure, diabetes, or heart disease.
According to the CDC, if you are a pregnant woman, reconsider traveling to Timor-Leste.
There is no specific treatment for chikungunya, but the CDC's Advisory Committee on Immunization Practices recommends the newly approved IXCHIQ® chikungunya vaccine for some travelers.
Jeri Beales, MSN RN with Destination Health Travel Clinic near Boston, MA, offered these comments, "With the CDC's new guidance for the IXCHIQ vaccine, all adults traveling to Timor-Leste should consider vaccination because infection with the virus has been linked to prolonged and severe fatigue and joint pain which can last months or years."
"The immunization experts at the CDC also recommend the IXCHIQ vaccine for some travelers going to countries that have reported cases in the previous five years."
"Travelers staying six months or longer are at the highest risk for infection, but vaccination can also be considered for those 65 years and older because they are more likely to have severe complications with an infection. If you plan to travel this summer, talk to your doctor or local travel health clinic to see if the IXCHIQ vaccine is recommended."
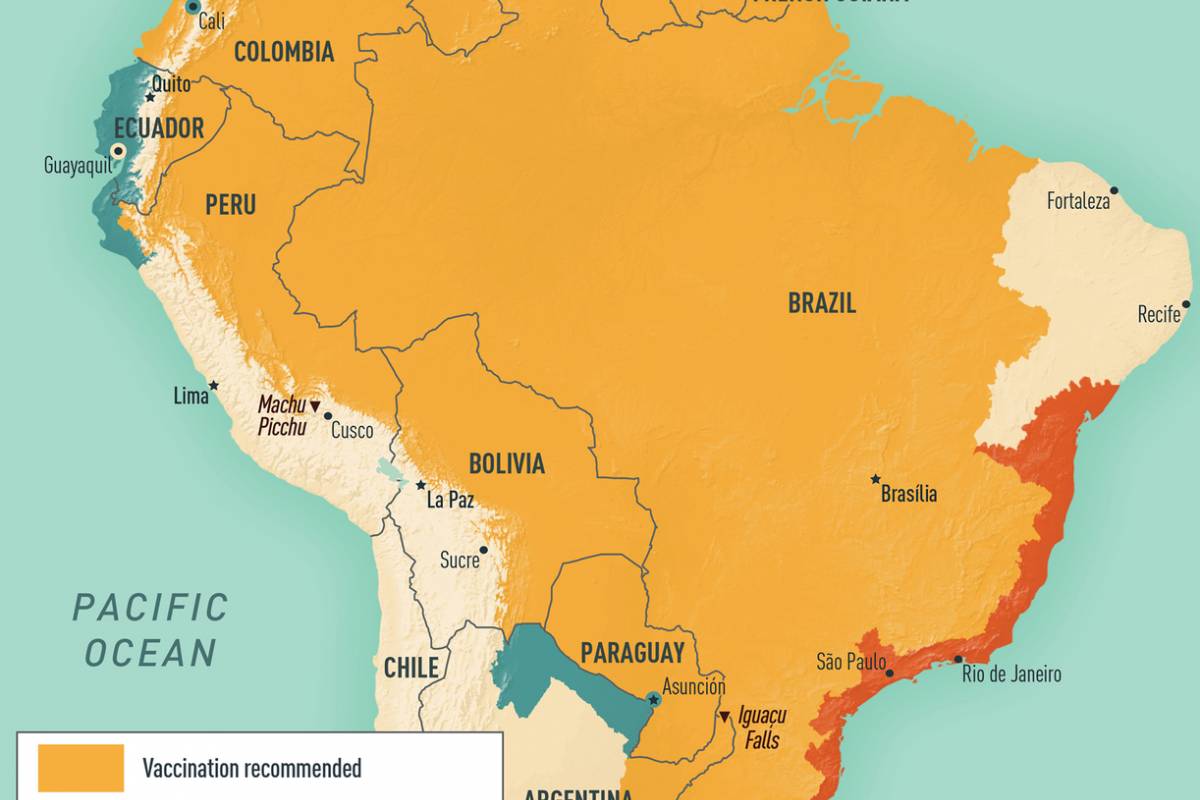
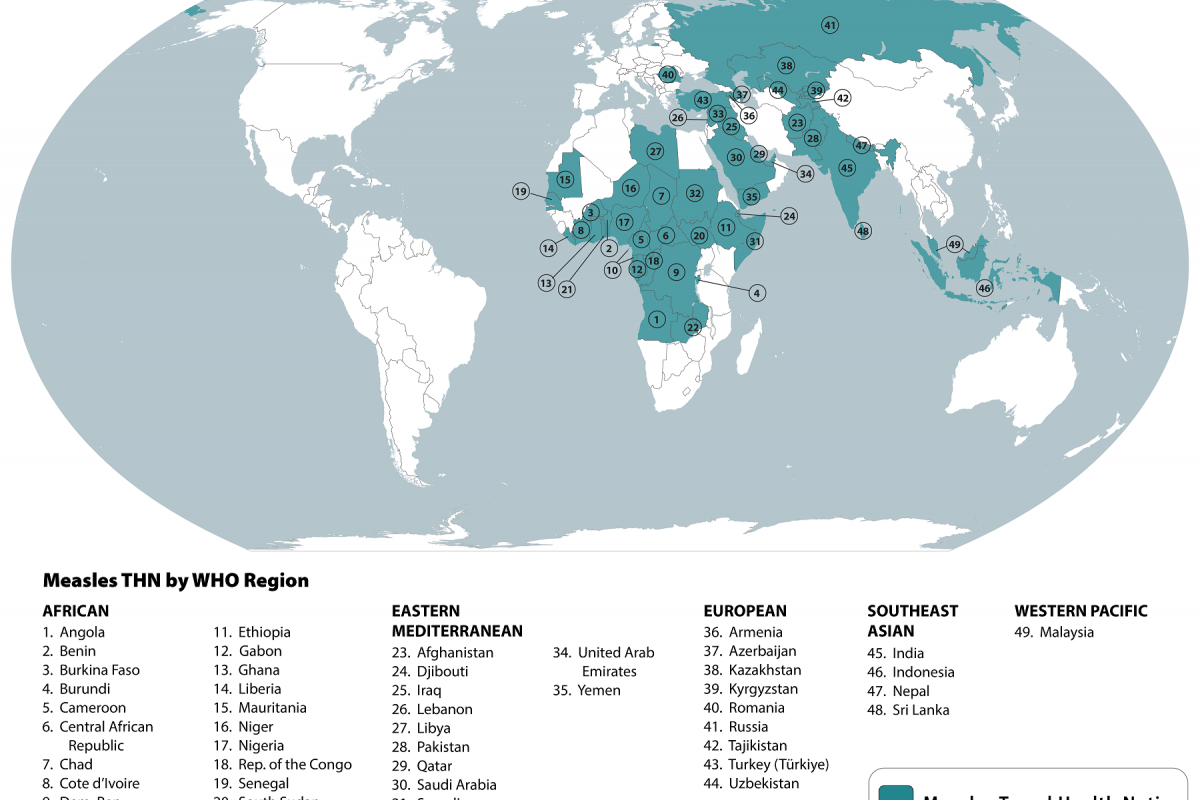
"Countries with outbreaks in the past five years span the globe, including regions of Central and South America, Africa, and Asia.
Timor-Leste is also experiencing dengue outbreaks and a resurgence of the Zika virus in the Ermera municipality in 2024.
As of late February 2024, local media reported 27 Zika cases and 460 dengue cases.