Houston and Miami May Become Yellow Fever Virus Gateways to America
With more than 3 billion passengers now flying on commercial flights each year, humans have become the primary agents for the spread of mosquito-borne viruses such as dengue, chikungunya, Zika, and yellow fever.
To reduce the spreading of these viruses, should the Miami and Houston International airports begin screening inbound international travelers for their yellow fever vaccination?
***Schedule your appointment today for recommended vaccines***
This potential passenger screening requirement is suggested by a new study from the World Health Organization (WHO).
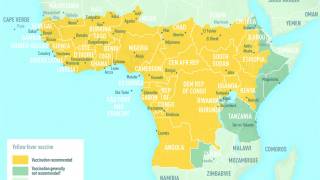
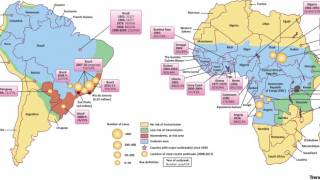
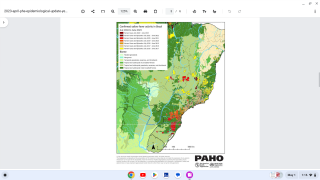
The study estimated that 923 million people lived in areas where yellow fever was endemic in 2016. And, because of a compatible environment, 472 cities were identified as suitable for future yellow fever outbreaks.
As of 2016, yellow fever vaccine had been introduced in routine infant immunization programmes in 35 of the 42 countries/territories at risk for yellow fever in Africa and the Americas.
In these 42 areas, vaccination coverage is estimated at 45 percent by the WHO.
In this study’s base scenario, 45.2 million travelers departed from yellow fever-endemic areas for international destinations in 2016.
Moreover, this study says 2.8 million people arrived in the United States during 2016 from endemic yellow fever virus countries.
But, USA airport customs did not verify any passenger’s immunization status.
In the USA, 9.5 million Americans live in urban areas that are ecologically suitable for yellow fever spreading mosquitoes, said this research.
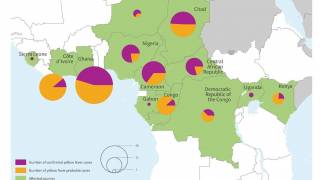
With Houston and Miami being prime targets given their tropical environment, and numerous visitors from South America and Africa, where yellow fever has reached outbreak status.
Brazil’s vaccination program for 23 million citizens has launched but appears to be proceeding slowly.
And, the WHO recently announced a massive program to immunize 1 billion Africans.
Yellow fever virus is a mosquito-borne flavivirus that causes yellow fever, an acute infectious disease that occurs in South America and sub-Saharan Africa. Most patients with yellow fever are asymptomatic.
But, among the 15 percent who develop severe illness, the yellow fever case fatality rate ranges up to 60 percent, says the WHO.
These researchers said that “Although the broader use of yellow fever vaccine by international travelers could limit dispersion of the virus and reduce the risk of urban epidemics, its use in non-epidemic settings must be carefully weighed against the risk of vaccine-associated neurological and viscerotropic events.”
“At a time when global yellow fever vaccine supplies are diminished, an epidemic in a densely populated city could have substantial health and economic consequences.”
“Hence, the global community needs to carefully re-examine existing yellow fever travel vaccination policies and practices to prevent urban epidemics.”
Yellow fever vaccine is recommended for people 9 months and older who are traveling to or living in areas at risk.
Sanofi Pasteur, the manufacturer of the only yellow fever vaccine (YF-Vax) licensed in the United States, announced during February 2018, that YF-Vax for civilian use is expected to be available from the manufacturer again by the end of 2018.
Until then, Sanofi Pasteur received approval from the US Food and Drug Administration to make Stamaril available to USA residents.
For most international travelers, a single dose of yellow fever vaccine provides long-lasting protection. However, some travelers may require a booster dose, says the WHO.
In the USA, selected pharmacies offer the STAMARIL and/or YF-VAX vaccines, as well as other travel vaccines.
Reactions to yellow fever vaccine are generally mild and include headaches, muscle aches, and low-grade fevers. There have been reports of serious events following yellow fever vaccination.
You are encouraged to report any vaccine side effects to the FDA or CDC.
The CDC Vaccine Price List provides the private sector prices and general information, and discounts can be found here.
The MUGK is financially supported by The Branco Weiss Fellowship - Society in Science and acknowledges funding from a Training Grant from the National Institute of Child Health and Human Development (T32HD040128) and the National Library of Medicine of the National Institutes of Health (R01LM010812, R01LM011965).
Our Trust Standards: Medical Advisory Committee