ACIP Vaccine Reviews Scheduled for June 2023

The U.S. CDC Advisory Committee on Immunization Practices (ACIP) confirmed it had scheduled a vaccine review meeting open to the public for June 21-23, 2023.
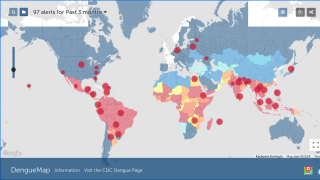
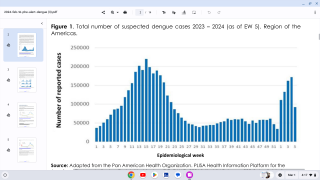
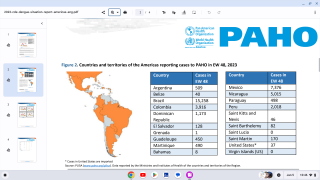
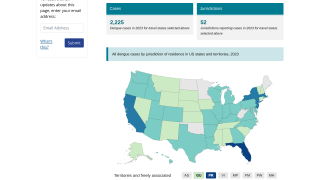
Conducted at the Centers for Disease Control and Prevention (CDC) in Atlanta, Georgia, this meeting's purpose is to review scientific data and vote on vaccines and candidate recommendations for Respiratory Syncytial Virus vaccines; Recommendations for adult Polio vaccinations; Flu shots for the 2023-2024 season, Chikungunya, COVID-19, Dengue, Meningococcal, and Mpox vaccines.
This meeting's agenda will be led by Dr. Grace Lee, the ACIP Chair.
ACIP recommendations are public health guidance for the safe use of vaccines and related biological products.
The non-binding recommendations include the age(s) when the vaccine should be given, the number of doses needed, the amount of time between doses, and precautions and contraindications.
Our Trust Standards: Medical Advisory Committee