Chikungunya Vaccine Candidate Completes BLA Submission to U.S. FDA

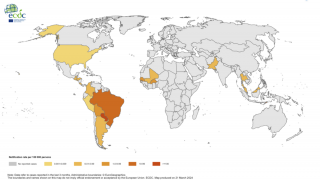
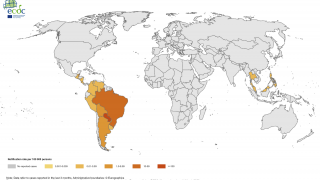
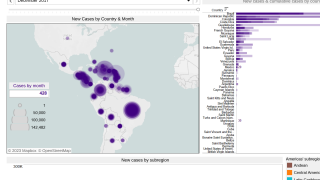
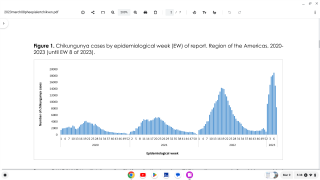
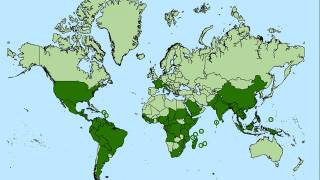
An innovative vaccine candidate for preventing chikungunya infections is fast approaching authorization in the U.S. The medical need for this vaccine is clear, as millions of chikungunya cases have been reported in the Americas and over 100 countries.
Furthermore, this mosquito-borne viral disease's related economic impact is considered significant.
France-based specialty vaccine company Valneva SE announced today that it had completed the rolling submission of the Biologics License Application (BLA) to the U.S. Food and Drug Administration (FDA) for its single-shot chikungunya vaccine candidate, VLA1553.
Valneva is seeking approval from the FDA for VLA1553 in adults aged 18 years and above.
The FDA will now review the filing for acceptance, determine priority review eligibility, and the action date that it targets to complete its evaluation.
This VLA1553 vaccine development program previously received FDA Fast Track and Breakthrough Therapy designations.
Juan Carlos Jaramillo, MD, Chief Medical Officer of Valneva, commented in a press release on December 23, 2022, "The completion of our BLA submission is extremely important as it takes us a step closer to potentially bringing a preventative solution to fight this debilitating disease."
"Chikungunya is a significant public health threat transmitted to humans by infected mosquitoes, and no vaccine or specific treatments for the disease are currently available."
"If the FDA approves the submission, our goal is to provide a tool to help curtail this growing, unmet medical need."
This BLA application follows the final pivotal Phase 3 data reported in March 2022 and the final lot-to-lot consistency results reported in May 2022.
A clinical study of VLA1553 in adolescents is ongoing in Brazil, which may support future regulatory submissions in this group if VLA1553 is initially approved in adults.
The Company also recently reported positive antibody persistence data with a 99% seroresponse rate 12 months after a single-dose vaccination.
VLA1553 was also granted PRIority MEdicine designation by the European Medicines Agency in 2020, and Valneva plans to make regulatory submissions for VLA1553 in Europe in the second half of 2023.
Chikungunya is a mosquito-borne viral disease caused by the chikungunya virus (CHIKV), a Togaviridae virus transmitted by Aedes mosquitoes.
Infections lead to symptomatic disease in 72-92% of humans after four to seven days following the mosquito bite.
While mortality with CHIKV is low, morbidity is high.
Clinical symptoms include acute onset of fever, debilitating joint and muscle pain, headache, nausea, rash, and chronic arthralgia.
CHIKV often causes sudden large outbreaks with high attack rates, affecting one-third to three-quarters of the population in areas where the virus is circulating.
As of December 23, 2022, there are no CHIKV preventive vaccines or effective treatments available, and, as such, chikungunya is considered a major public health threat.
If approved, Valneva intends to commercialize VLA1553, leveraging its existing manufacturing and commercial operations.
Other chikungunya vaccine development news is posted at PrecisionVaccinations.com.
Our Trust Standards: Medical Advisory Committee