California Needs a Lyme Disease Vaccine
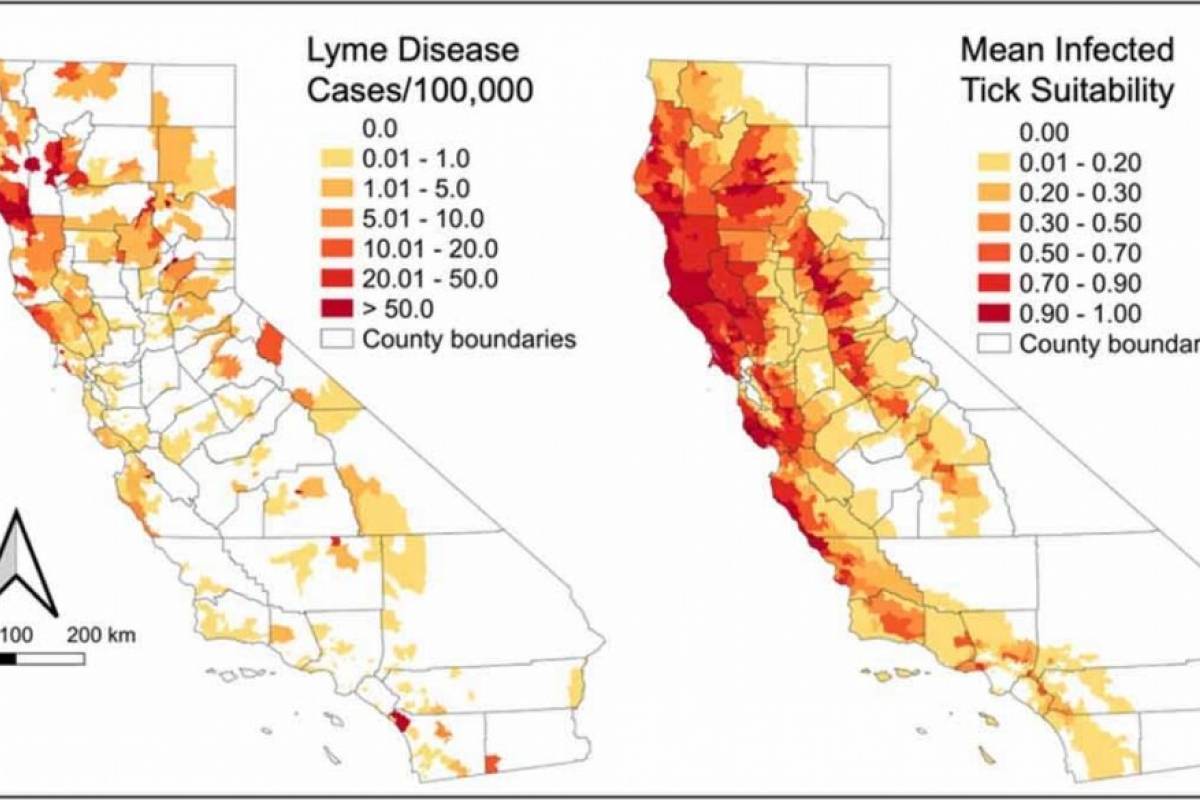
Researchers at UC Santa Barbara recently discovered the ecology of the small mammals upon which ticks feed, helping explain the rates of human Lyme disease, at least in California.
As a result, scientists and health officials may be able to predict future disease risk by studying the response of these animals, and their tick parasites, to changing climate and land use.
"This study is unique because it tries to quantify the links connecting climate to mammals to ticks to humans, which requires different types of data, research techniques, and academic backgrounds," said co-author Sam Sambado, a doctoral student in the Department of Ecology, Evolution, and Marine Biology, in a UCSB press release on November 21, 2022.
Lyme disease is primarily caused by the bacterium Borrelia burgdorferi by tick bites.
However, the western blacklegged tick isn't born with the bacterium.
Instead, it can only contract the pathogen by feeding on an infected host, which serves as a reservoir for the microbe.
While no U.S. FDA-approved vaccine prevents Lyme disease infections, a candidate is making significant progress.
Lyme Disease vaccine candidate VLA15 is a multivalent recombinant protein vaccine targeting the outer surface protein A (OspA) of Borrelia, designed for prophylactic, active immunization against Lyme disease to protect people against most human pathogenic Borrelia species.
OspA is one of the most dominant surface proteins expressed by the bacteria when present in a tick.
Initially developed by Valneva SE, the VLA15 Lyme disease vaccine candidate confers protection by raising antibodies that prevent Borrelia from migrating from ticks to humans.
The VLA15 vaccine program was granted Fast Track designation by the FDA in July 2017.
And in April 2020, Valneva and Pfizer, Inc. announced a development and commercialization collaboration for VLA15.
As of November 2022, VLA15 remains the only active Lyme disease vaccine program in human clinical development.
This study's findings appeared in the journal Environmental Research Letters on November 9, 2022.
Our Trust Standards: Medical Advisory Committee























