Chikungunya Virus-like Particle Vaccine Candidate Induced Robust Neutralizing Antibody Immune Responses

Maryland-based Emergent BioSolutions Inc. announced today that two-year persistence data evaluating the safety and immunogenicity of the company's investigational chikungunya virus virus-like particle (CHIKV VLP) vaccine candidate PXVX0317 were published in The Lancet Infectious Diseases.
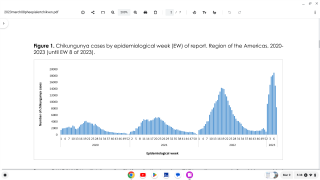
The results from its phase 2 clinical study were published on June 13, 2022, and concluded a single 40 μg injection of adjuvanted PXVX0317 was well tolerated and induced a robust and durable serum neutralizing antibody immune response against CHIKV for up to 2 years.
"Emergent is pleased that results from our CHIKV VLP phase 2 study have been published and shared with the scientific community in The Lancet Infectious Diseases, a highly esteemed peer-reviewed journal," said Chris Cabell, M.D., chief medical officer and SVP clinical development at Emergent BioSolutions in a press release issued on June 21, 2022.
"Publication of this body of work highlights the importance of developing ways to address Chikungunya disease, a public health threat for which no vaccine or treatment exists."
This randomized, double-blind, parallel-group, phase 2 study was conducted at three U.S. sites with 415 CHIKV-naïve adults aged 18–45 years.
The primary endpoint was the geometric mean titer of the anti-CHIKV neutralizing antibody on day 57 (28 days after the last vaccination). Safety was also assessed.
As of June 21, 2022, the U.S. FDA has not approved a Chikungunya vaccine.
Additional chikungunya vaccine development news is posted at Vax-Before-Travel.
Note: Emergent's announcement was manually curated for mobile readers.
Our Trust Standards: Medical Advisory Committee