100-Year-Old Vaccine Remains Broadly Protective in Newborns

Researchers at the Precision Vaccines Program at Boston Children’s Hospital recently partnered with the Expanded Program on Immunization Consortium, an international team studying early life immunization, to collect and comprehensively profile blood samples from newborns immunized with the century-old Bacille Calmette-Guérin (BCG) vaccine, using a powerful “big data” approach.
Published by the peer-review journal Cell Reports on May 3, 2022, this study found the tuberculosis (TB)-focused BCG vaccine protected newborns and infants against multiple bacterial and viral infections unrelated to TB.
And BCG vaccinations at birth changed metabolite and lipid profiles in newborns’ blood plasma in a pattern distinct from those in the delayed-vaccine group. These changes correlated with innate immune system responses.
The researchers had similar findings when they tested for BCG in cord blood samples from a cohort of newborns in Boston, MA, from a separate NIH/NIAID-funded Human Immunology Project Consortium study in The Gambia and Papua New Guinea.
“A growing number of studies show that BCG vaccination protects against unrelated infections,” says Ofer Levy, MD, Ph.D., director of the Precision Vaccines Program and the study’s senior investigator, in a related press release.
"BCG is an ‘old school’ vaccine — it’s made from a live, weakened germ — but live vaccines like BCG seem to activate the immune system in a very different way in early life, providing broad protection against a range of bacterial and viral infections."
“It’s critical that we learn from BCG to understand better how to protect newborns."
"There’s much work ahead to better understand that and use that information to build better vaccines for infants.”
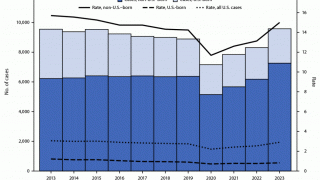
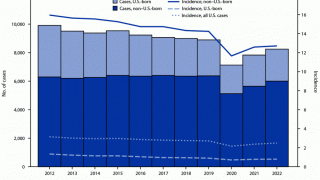
In the U.S., the BCG vaccine is not integrated into the children's vaccination schedule.
And it is not generally recommended for use because of the low risk of infection with Mycobacterium tuberculosis, the variable effectiveness of the vaccine against adult pulmonary TB, and the vaccine’s potential interference with tuberculin skin test reactivity says the U.S. CDC.
However, in the U.K., the BCG vaccine is given when a child is at increased risk of coming into contact with TB.
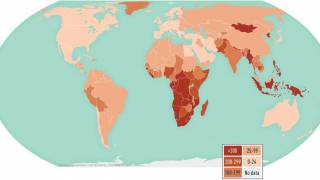
Eligible babies include all newborns whose parent/grandparent/ were born in a country where the annual incidence of TB is 40 per 100,000 or greater; or newborn babies living in areas of the U.K. where the annual incidence of TB is 40 per 100,000 or greater.
Additionally, the evaluation of screening for Severe Combined Immunodeficiency (SCID) to the routine newborn screening test at five days of age made it necessary to move the BCG vaccination to when a SCID screening outcome will be available which may be available from around day 14 to 17 after birth.
This change, announced by the UK Health Security Agency on May 6, 2022, ensured that babies with SCID are not given the live attenuated BCG vaccine, which is contraindicated.
Notes: The study was supported by the NIAID (U19AI118608, U01 AI124284), the Precision Vaccines Program at Boston Children’s Hospital, and the Mueller Health Foundation. In addition, Dr. Levy is a named inventor of several Boston Children's Hospital patents relating to human microphysiologic assay systems and vaccine adjuvants. Study coauthors Scott McCulloch and Greg Michelotti are employees of Metabolon Inc. The other authors declare no competing financial interests.
This study and press release were edited for clarity and manually curated for mobile readership.
Our Trust Standards: Medical Advisory Committee