Zika Vaccine Candidate Found Effective and Safe

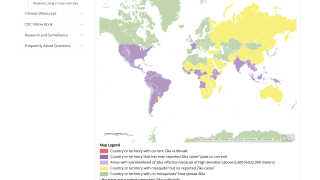
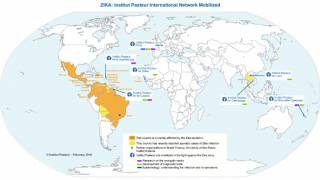
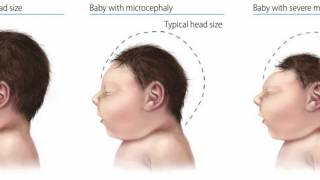
Since 2015, the emergence of epidemic Zika virus transmission has been linked to unparalleled reports of serious neurological disorders and devastating congenital disabilities such as microcephaly and other congenital malformations.
Despite low transmission rates in the past 3 years, the emergence of new Zika outbreaks remains a looming threat, stated an editorial published by The Lancet on May 18, 2021.
Furthermore, as of May 24, 2021, the U.S. FDA has not Approved any Zika vaccine candidate.
However, in a separate study published by the Lancet, Japan-based Takeda Vaccines's TAK-426 vaccine candidate was reported well tolerated, with an acceptable safety profile, and immunogenic in both flavivirus-naive and flavivirus-primed adults.
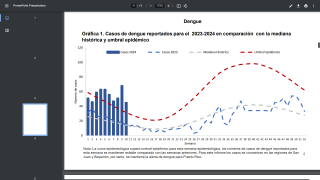
This two-part, multicentre, observer-blind, randomized, placebo-controlled, phase 1 trial was done at seven medical clinics in the USA and two in Puerto Rico. Between November 2017 and October 24, 2018, 271 people were enrolled (125 flavivirus-naive and 146 flavivirus-primed participants).
All TAK-426 vaccine doses were well tolerated with no deaths, no vaccine-related serious adverse events, and similar rates of mainly mild to moderate adverse events. TAK-426 elicited dose-dependent increases in antibody GMTs in both flavivirus-naive and flavivirus-primed participants. 28 days after dose 2, plaque-reduction neutralization test GMTs in flavivirus-naive participants were 1130 (95% CI 749–1703) in the 2 μg TAK-426 group, 1992 (1401–2833) in the 5 μg TAK-426 group, and 3690 (2677–5086) in the 10 μg TAK-426 group.
Based on the safety and immunogenicity profiles of all TAK-426 vaccine doses assessed, the 10 μg TAK-426 dose was selected for further clinical development.
This study's funding originated from Takeda Vaccines and the US Biomedical Advanced Research and Development Authority.
Osaka, Japan-based Takeda is a patient-focused, values-based, R&D-driven global biopharmaceutical company committed to bringing Better Health and a Brighter Future to people worldwide.
Our Trust Standards: Medical Advisory Committee