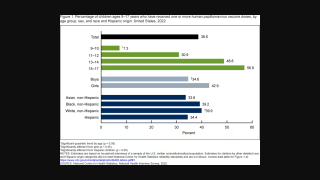
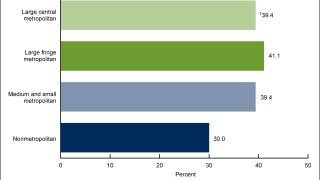
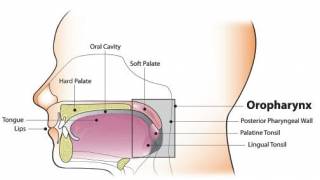
HPV Vaccination While Pregnant Found Not Associated with Adverse Birth Outcomes

The JAMA Network Open published a human papillomavirus (HPV) vaccine study on April 5, 2021, that found women who received the 9-valent human papillomavirus (9vHPV) vaccine during early pregnancy did not experience an increase in miscarriages or adverse birth outcomes.
And among live births, 9vHPV vaccine exposures during or around the time of pregnancy were not associated with adverse birth outcomes.
This cohort study analyzed data from seven participating health systems in the Vaccine Safety Datalink. The cohort comprised pregnancies among women aged 12 to 28 years that ended between October 26, 2015, and November 15, 2018. Singleton pregnancies that ended in a live birth, stillbirth, or SAB were included.
The data used included more than 800 live births with during-pregnancy or peripregnancy 9vHPV vaccine exposures and nearly 700 live births with follow-up to evaluate for congenital disabilities.
These researchers concluded by saying, 'This study found that 9vHPV vaccine exposures during or around the time of pregnancy were uncommon and not associated with spontaneous abortions or selected adverse birth outcomes.'
'Given the continual need for catch-up 9vHPV vaccination in women up to age 26 years, along with the expanded use of 9vHPV vaccination in women up to age 45 years, the findings from this study can inform counseling after inadvertent 9vHPV vaccine exposures during or around the time of pregnancy.' Note: This study's sample size was limited.
A list of approved HPV vaccines is rendered on this webpage.
This study was funded by a contract (200-2012-53526-0006) with the CDC. Dr. Sheth was supported in part by K07CA230234 from the National Cancer Institute of the National Institutes of Health. No industry conflicts of interest were disclosed.
Our Trust Standards: Medical Advisory Committee