Single-Shot Chikungunya Vaccine Candidate Launches Phase 3 Clinical Lot Consistency Study
France-based Valneva SE announced that it had initiated the clinical lot-to-lot consistency Phase 3 study for its single-shot chikungunya vaccine candidate, VLA1553. This is the only chikungunya vaccine candidate in Phase 3 clinical trials.
This study aims to show the vaccine's manufacturing consistency by demonstrating that three consecutively manufactured lots elicit equivalent immune responses measured by neutralizing antibody titers on Day 29 after vaccination. Lyophilized VLA1553 will be administered as a single intramuscular immunization.
This study will run parallel to the ongoing, pivotal Phase 3 study, VLA1553-301, which includes the determination of seroprotection based on an immunological surrogate.
Juan Carlos Jaramillo, M.D., Chief Medical Officer of Valneva, commented in a press statement issued on February 22, 2021, “The chikungunya virus continues to represent a major public health threat, and we are working as fast as we can to bring a preventive solution to those who need it most. We want to thank our employees, partners, and study participants for making this trial possible despite the ongoing COVID-19 pandemic.”
"This study initiation signifies another important step in the development of VLA1553 towards licensure."
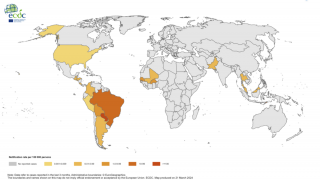
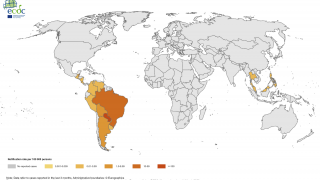
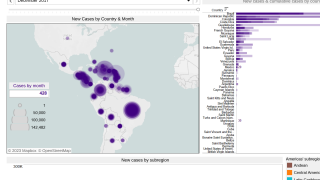
The sponsor of the first chikungunya vaccine Biologics License Application (BLA) to be approved in the U.S. (Butantan Institute) will be eligible to receive a Priority Review Voucher. According to a 2020 report from the U.S. Government Accountability Office (GAO) about the priority review voucher (PRV) program, “all seven drug sponsors GAO spoke with stated that PRVs were a factor in drug development decisions—six sponsors said they were one of several factors, while one sponsor said they were pivotal in its development of a drug.”
Valneva SE has operations in Austria, Sweden, the United Kingdom, France, Canada, and the U.S. with over 500 employees.
Our Trust Standards: Medical Advisory Committee