Ebola Vaccine Stockpiled for Immediate Delivery

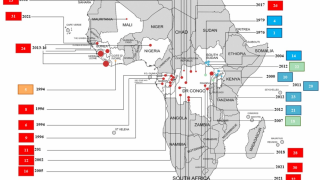
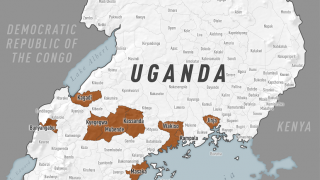
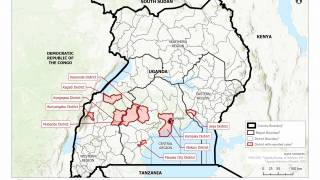
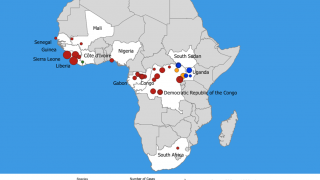
UNICEF announced international health and humanitarian organizations had established a global Ebola vaccine stockpile to ensure timely outbreak response on January 12, 2021. This vaccine stockpile will enable countries to contain future Ebola epidemics by ensuring access to vaccines for populations at risk during outbreaks.
The injectable single-dose Ervebo Ebola vaccine is manufactured by Merck, Sharp & Dohme Corp. and developed with financial support from the US government. The European Medicines Agency licensed the Ervebo vaccine in November 2019. The vaccine is now prequalified by the WHO and licensed by the U.S. FDA and eight African countries.
Prior to licensure, Ervebo was administered to more than 350,000 people under the “compassionate use” protocol.
Dr. Tedros Adhanom Ghebreyesus, WHO Director-General, stated in a press release, “Ebola vaccines have made one of the most feared diseases on earth preventable. This new stockpile is an excellent example of solidarity, science, and cooperation between international organizations and the private sector to save lives.”
The vaccine stockpile is stored in Switzerland and ready to be shipped to countries for emergency response. The decision to allocate the vaccine will be made within 48 hours of receiving a country's request; vaccines will be made available together with ultra-cold chain packaging by the manufacturer for shipment to countries within 48 hours of the decision.
Our Trust Standards: Medical Advisory Committee